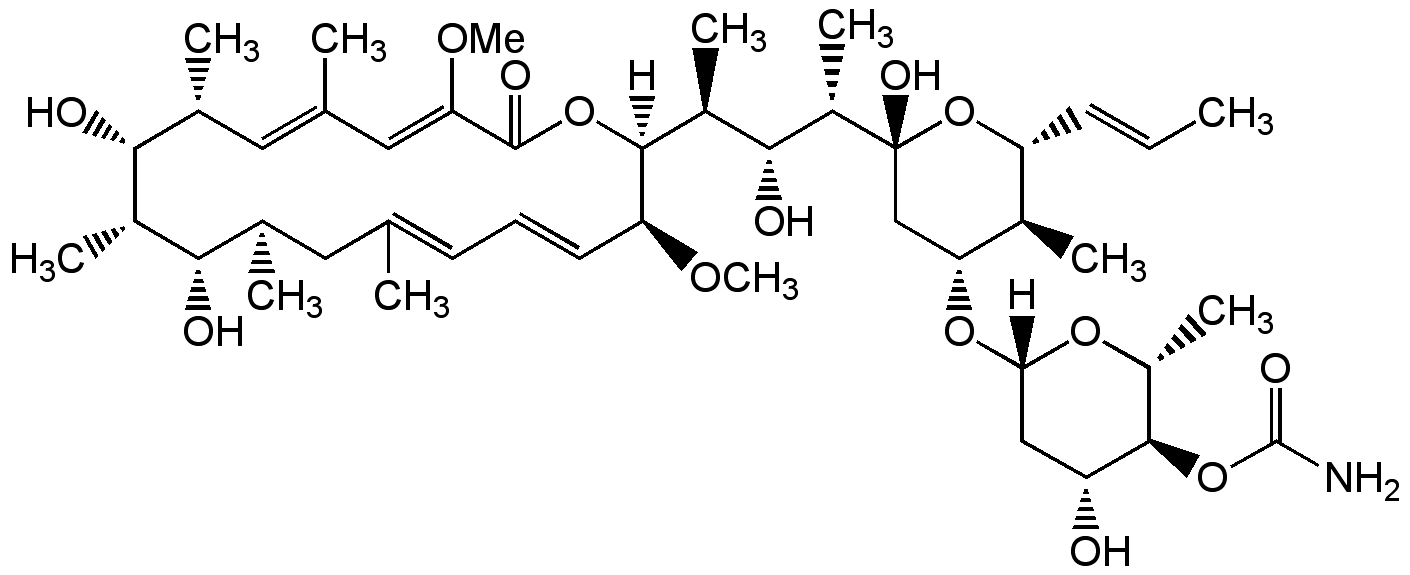
Concanamycin B
Product Code:
BVT-0253
BVT-0253
Regulatory Status:
RUO
RUO
Shipping:
20°C
20°C
Storage:
-20°C
-20°C
No additional charges, what you see is what you pay! *
| Code | Size | Price |
|---|
| BVT-0253-C025 | 25 ug | £95.00 |
Quantity:
| BVT-0253-C100 | 100 ug | £170.00 |
Quantity:
| BVT-0253-C500 | 500 ug | £490.00 |
Quantity:
| BVT-0253-M001 | 1 mg | £740.00 |
Quantity:
Prices exclude any Taxes / VAT
Stay in control of your spending. These prices have no additional charges, not even shipping!
* Rare exceptions are clearly labelled (only 0.14% of items!).
* Rare exceptions are clearly labelled (only 0.14% of items!).
Multibuy discounts available! Contact us to find what you can save.
This product comes from: Switzerland.
Typical lead time: 10-14 working days.
Contact us for more accurate information.
Typical lead time: 10-14 working days.
Contact us for more accurate information.
- Further Information
- Documents
- References
- Show All
Further Information
Alternate Names/Synonyms:
8-Deethyl-8-methylconcanamycin A
Appearance:
White to off-white solid.
CAS:
81552-33-2
Class:
6.1
EClass:
32160000
Form (Short):
liquid
GHS Symbol:
GHS06
Handling Advice:
Protect from light when in solution.
Hazards:
H300, H310, H319, H332
InChi:
InChI=1S/C45H73NO14/c1-13-15-33-27(6)36(57-37-21-32(47)42(31(10)56-37)59-44(46)52)22-45(53,60-33)30(9)40(50)29(8)41-34(54-11)17-14-16-23(2)18-25(4)38(48)28(7)39(49)26(5)19-24(3)20-35(55-12)43(51)58-41/h13-17,19-20,25-34,36-42,47-50,53H,18,21-22H2,1-12H3,(H2,46,52)/b15-13+,17-14+,23-16-,24-19+,35-20-/t25-,26-,27-,28+,29+,30+,31-,32-,33-,34?,36-,37+,38+,39-,40-,41+,42-,45-/m1/s1
InChiKey:
AQXXGIBOZQZSAT-UZYXLCJWSA-N
Long Description:
Chemical. CAS: 81552-33-2. Formula: C45H73NO14. MW: 852.1. Isolated from Streptomyces sp. Antibiotic. Exhibits similar activity as concanamycin A (Prod. No. BVT-0237) and concanamycin C (Prod. No. BVT-0254). Specific vacuolar-type H+-ATPase inhibitor. Inhibitor of autophagic degradation by rising lysosomal pH and thus inactivating the lysosomal acid hydrolases. Suppresses bone resorption. Inhibits proliferation of mouse splenic lymphocytes. Antifungal and larvicidal.
MDL:
MFCD01731919
Molecular Formula:
C45H73NO14
Molecular Weight:
852.1
Package Type:
Plastic Vial
PG:
III
Precautions:
P261, P262, P280, P301, P310, P302, P350, P312
Product Description:
Antibiotic. Exhibits similar activity as concanamycin A (Prod. No. BVT-0237) and concanamycin C (Prod. No. BVT-0254). Specific vacuolar-type H+-ATPase inhibitor. Inhibitor of autophagic degradation by rising lysosomal pH and thus inactivating the lysosomal acid hydrolases. Suppresses bone resorption. Inhibits proliferation of mouse splenic lymphocytes. Antifungal and larvicidal.
Purity:
>98% (HPLC)
Signal word:
Danger
SMILES:
[H][C@@]1(C[C@@H](O)[C@H](OC(N)=O)[C@@H](C)O1)O[C@@H]1C[C@@](O)(O[C@H](C=CC)[C@H]1C)[C@@H](C)[C@H](O)[C@H](C)[C@]1([H])OC(=O)C(OC)=CC(C)=C[C@@H](C)[C@@H](O)[C@@H](C)[C@@H](O)[C@H](C)CC(C)=CC=C[C@@H]1OC
Solubility Chemicals:
Soluble in DMSO, chloroform, acetonitrile or methanol; insoluble in water.
Source / Host:
Isolated from Streptomyces sp.
Transportation:
Excepted Quantity
UN Nummer:
UN 3462
UNSPSC Category:
Natural Products/Extracts
UNSPSC Number:
12352200
Use & Stability:
Stable for at least 1 year after receipt when stored at -20°C. After reconstitution protect from light at -20°C.
Documents
References
Structures of concanamycins B and C: H. Kinashi, et al.; J. Antibiot. 35, 1618 (1982) | Isolation and characterization of concanamycins A, B and C: H. Kinashi, et al.; J. Antibiot. (Tokyo) 37, 1333 (1984) | Involvement of the vacuolar H(+)-ATPases in the secretory pathway of HepG2 cells: M. Yilla, et al.; J. Biol. Chem. 268, 19092 (1993) | Inhibitory effect of modified bafilomycins and concanamycins on P- and V-type adenosintriphosphatase: S. Drose, et al.; Biochemistry 32, 3902 (1993) | Characterization of the ATPase activity of P-glycoprotein from multidrug-resistant Chinese hamster ovary cells: F.J. Sharom, et al.; Biochem. J. 308 (Pt2), 381 (1995) | Concanamycin B inhibits the expression of newly synthesized MHC class II molecules on the cell surface: K. Ito, et al.; J. Antibiot. 48, 488 (1995) | Concanamycin B, a vacuolar H(+)-ATPase specific inhibitor suppresses bone resorption in vitro: J.T. Woo, et al.; Biol. Pharm. Bull. 19, 297 (1996) | In vivo rapid reduction of alloantigen-activated CD8+ mature cytotoxic T cells by inhibitors of acidification of intracellular organelles, prodigiosin 25-C and concanamycin B: M. H. Lee, et al.; Immunology 99, 243 (2000) | Vacuolar H+-ATPase inhibitors overcome Bcl-xL-mediated chemoresistance through restoration of a caspase-independent apoptotic pathway: Y. Sasazawa, et. al.; Cancer Sci. 100, 1460 (2009) | Inhibitors of the V0 subunit of the vacuolar H+-ATPase prevent segregation of lysosomal- and secretory-pathway proteins: J. A. Sobota, et. al.; J. Cell Sci. 122, 3542 (2009) | Inhibitors of V-ATPase: old and new players: M. Huss, et. al.; J. Exp. Biol. 212, 341 (2009)